No way we could publish this story without a photo of Jell-O
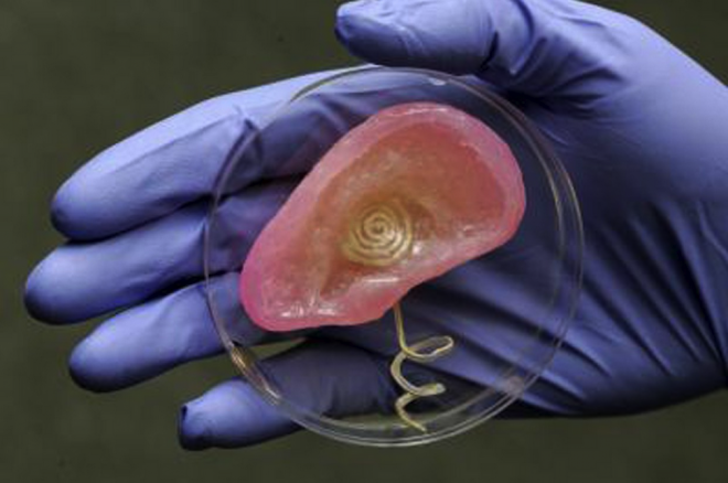
Standford researchers have come up with a new gel that could someday allow people to 3d print electronic circuits. According to the team, this electrically conductive gel with unimagined electrical performance is supposedly very easy to make and can be printed out on an inkjet printer (and obviously a 3D printer).
According to the Stanford University News, the researchers see it being used in things as widespread as futuristic energy storage devices, medical sensors, biofuel cells and medical probes. Until now, the characteristics that makeup this magical gel have been difficult to manufacture. But this new substance is made up of commercially available ingredients which go into a water solution.
Credit for creating the gel goes to Stanford chemical engineering Associate Professor Zhenan Bao, materials science and engineering Associate Professor Yi Cui and members of their labs.
The Jello-like material is something like a conducting hydrogel, which feels and behaves like biological tissues, but conducts electricity like a metal or semiconductor. These characteristics, along with its large surface area and its electrical capabilities, make it a great medium that will allow biological systems to communicate with technological hardware.
Here’s how it works:
Bao and Cui made the gel by binding long chains of the organic compound aniline together with phytic acid, found naturally in plant tissues. The acid is able to grab up to six polymer chains at once, making for an extensively cross-linked network.
“There are already commercially available conducting polymers,” said Bao, “but they all form a uniform film without any nanostructures.”
In contrast, the new gel’s cross-linking makes for a complex, sponge-like structure. The hydrogel is marked with innumerable tiny pores that expand the gel’s surface area, increasing the amount of charge it can hold, its ability to sense chemicals, and the rapidity of its electrical response.
Still, the gel can be easily manipulated. Because the material doesn’t solidify until the last step of its synthesis, it can be printed or sprayed as a liquid and turned into a gel after it’s already in place – meaning that manufacturers should be able to construct intricately patterned electrodes at low cost.
“You can’t print Jell-O,” said Cui. “But with this technique, we can print it and make it Jell-O later.”
The material’s unusual structure also gives the gel what Cui referred to as “remarkable electronic properties.”
Most hydrogels are tied together by a large number of insulating molecules, reducing the material’s overall ability to pass electrical current. But phytic acid is a “small-molecule dopant” – meaning that when it links polymer chains, it also lends them charge. This effect makes the hydrogel highly conductive.
We’ve got much of the technology of 3D printing down already. Now the spotlight is shining on the materials we print with, and none are more exciting than the biomaterials coming out of labs now.
The research appears this month in the journal PNAS.
Source: Stanford University News
Stanford develops revolutionary, highly-conductive, 3D-printable, biological Jell-O-like material
by Mark Fleming